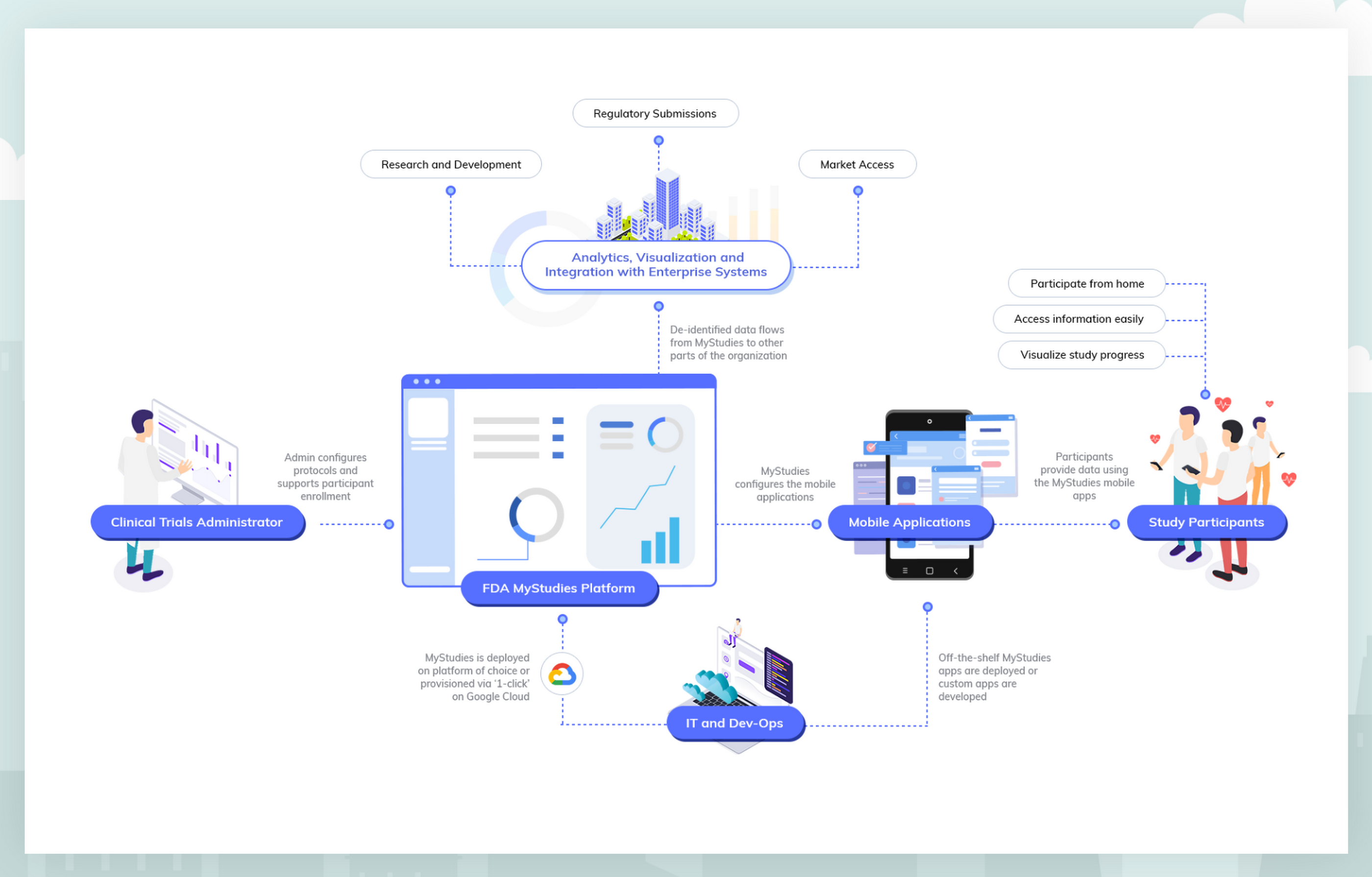
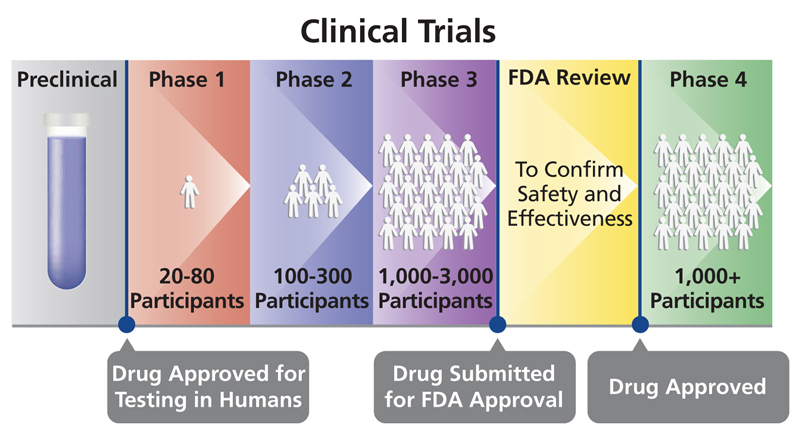
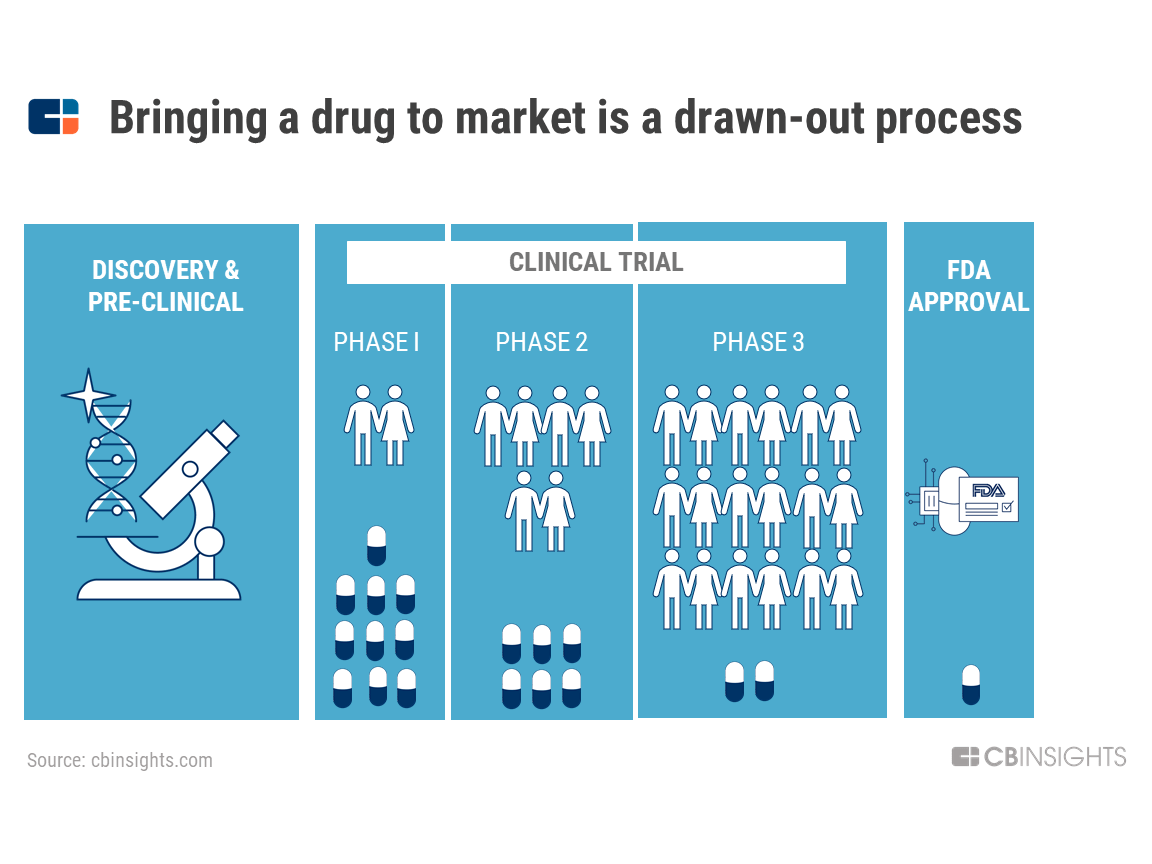
Incorporating Site-less Clinical Trials Into Drug Development: A Framework for Action - Clinical Therapeutics

FDA's own documents reveal agency's lax, slow, and secretive oversight of clinical research | Science | AAAS

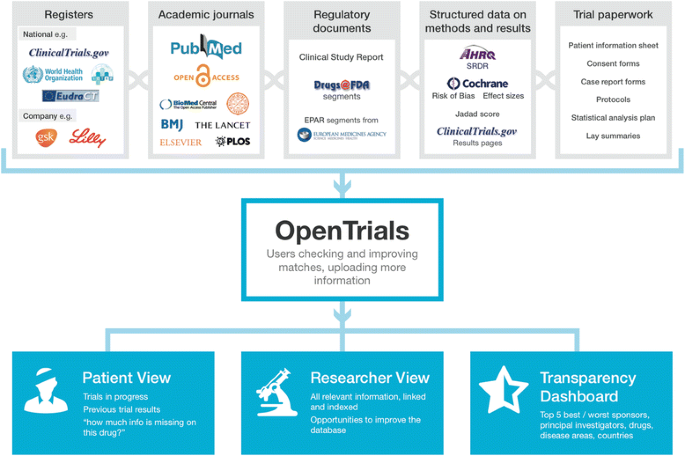
Clinical Trial Transparency: Ensuring Compliance with the New FDA Guidelines | BrackenData Clinical Trial Intelligence

Stevens-Johnson syndrome/toxic epidermal necrolysis in patients treated with immune checkpoint inhibitors: A safety analysis of clinical trials and FDA pharmacovigilance database - eClinicalMedicine